A group of Russian researchers from the Skolkovo Institute of Science and Technology (Skoltech) published a comprehensive analysis of the use of polycyclic aromatic hydrocarbons (PAHs) in energy storage technologies. The authors of the work — Ilya Chepkasov, Stanislav Evlashin, and Alexander Kvashnin — systematized the achievements of the past decade in using this class of organic compounds to create anodes and cathodes for metal-ion batteries.

Polycyclic aromatic hydrocarbons (PAHs) are of interest because of their special properties. These properties allow them to easily transfer electrons and admit ions into their structure. The behavior of PAHs in chemical reactions depends on their energy levels. If the energy level of the lowest unoccupied molecular orbital (LUMO) is low, PAHs are easily reduced, which is useful for anode applications. If the energy level of the highest occupied molecular orbital (HOMO) is low, PAHs are easily oxidized, which is useful for cathodes.
We have shown that ordinary aromatic compounds, such as naphthalene or anthracene, can perform no worse than, and in some cases even better than, expensive synthetic materials.
The study explains how special substances (PAHs) are used in different types of batteries: lithium-ion, sodium-ion, aluminum-ion, and dual-ion. Special attention is paid to the pre-metallization method. It helps reduce energy losses and improve battery performance from the very beginning.
This technology solves the fundamental problem of the first battery operating cycles, when a significant portion of metal ions is spent on forming a protective film on the anode.
For the first time within a single review, the authors examine the integration of machine learning for accelerated design of PAH materials. High-throughput screening makes it possible to predict the electrochemical properties of compounds before their synthesis.
Machine learning is now used everywhere in battery development. Over the past couple of years, it has turned from an experimental method into a standard working tool. In just one year, an enormous number of significant papers on this topic have been published: on AI-based molecule search, new cathodes, electrolyte selection and their behavior, accelerated analysis of materials for solid-state and lithium-sulfur batteries. And these are not just studies — convenient models and even entire AI-based software programs are already appearing.
The market context reinforces the relevance of the development. Global demand for lithium-ion batteries is projected at 4.7 TWh by 2030, compared with 700 GWh in 2022. Rising prices for metallic lithium are stimulating the search for alternatives with more affordable charge-carrying metals.
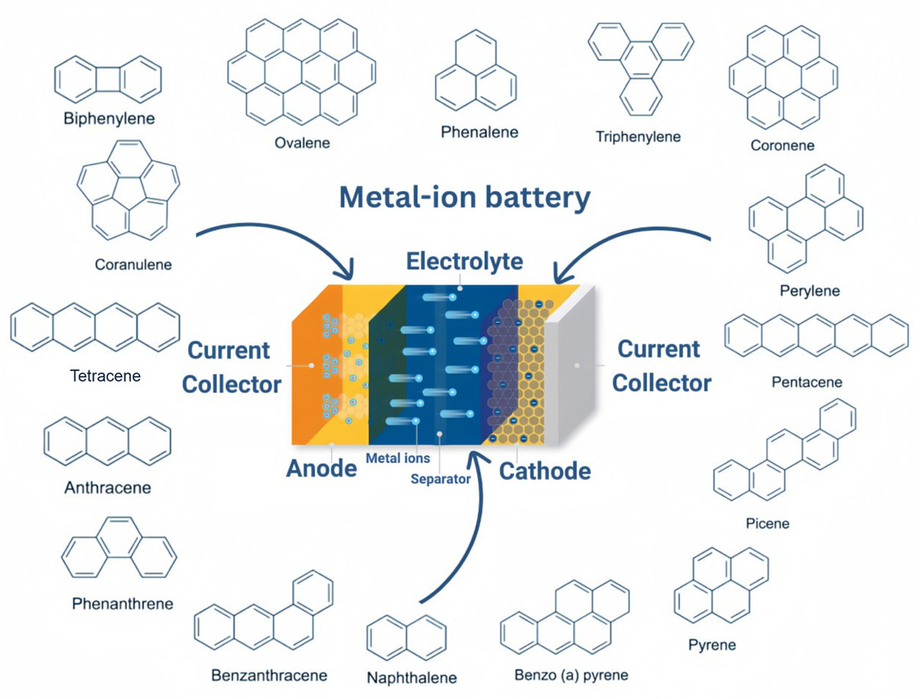
PAH materials compete with traditional inorganic electrodes. Graphite anodes provide a specific capacity of about 372 mAh/g, whereas some PAHs demonstrate values above 500 mAh/g. Transition metal oxides for cathodes are toxic and expensive to produce, while organic compounds are biodegradable. Silicon anodes suffer from volume expansion during cycling — a problem less pronounced in molecular structures. Sodium-ion technologies based on PAHs solve the problem of lithium scarcity. Chemical regeneration technology with pyrene-lithium complexes makes it possible to restore spent batteries to 100% of their original capacity. Challenges remain: the solubility of compounds in electrolytes and the insufficient redox activity of some molecules require further research into functionalization and structural optimization.
Read more on the topic:
-
 Nornickel Develops Lithium-Sulfur Batteries with Palladium for Electric Vehicles
Nornickel Develops Lithium-Sulfur Batteries with Palladium for Electric Vehicles -
 Now the cold won't kill the battery: TPU scientists, together with colleagues from China, have created a storage device for extreme conditions
Now the cold won't kill the battery: TPU scientists, together with colleagues from China, have created a storage device for extreme conditions -
 33 million battery cells per year: a mega-cluster for battery production is being deployed in Moscow
33 million battery cells per year: a mega-cluster for battery production is being deployed in Moscow
















